Novel low-bandgap donor–acceptor thiophene-phenylene co-oligomers for light-emitting semiconductor devices
By Lucia Feriancov´a, Dmitry O. Balakirev, Roman S. Fedorenko, Alexey V. Kuevda,Vasiliy A. Trukhanov, Eugenia A. Svidchenko, Nikolay M. Surin, Svetlana M. Peregudova, Petr V. Dmitryakov, Nikita O. Dubinets, Yuriy V. Fedorov,Martin Putala, Sergey A. Ponomarenko, Dmitry Yu. Paraschuk, Yuriy N. Luponosov
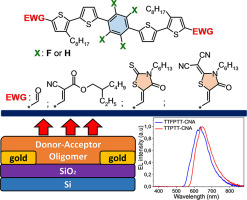
Thiophene-phenylene co-oligomers (TPCOs) have shown their high potential for organic light-emitting devices because of their high luminescence and efficient charge transport. However, unsubstituted TPCOs have relatively wide optical bandgaps and the high-lying lowest unoccupied molecular orbital (LUMO) energies so that efficient electron transport is a challenge. Electron-withdrawing groups (EWGs) and fluorinated fragments embedded into the TPCO molecule structure could result in the lower LUMO energy and narrower optical bandgap. Here, we report the synthesis of two novel TPCOs series with either phenylene or perfluorinated phenylene central core and end-capped with various EWGs (aldehyde, 2-ethylhexyl cyanoacetate, hexyl rhodanine and dicyanorhodanine) and with long alkyl terminal and side chains increasing the solubility. All the oligomers synthesized were found to be thermally stable and crystalline materials with relatively low LUMO energies (down to −3.50 eV), narrow bandgaps (down to 1.9 eV), and efficient photoluminescence in the green – deep red spectral regions both in solution and solid-state. The TPCOs with 2-ethylhexyl cyanoacetate EWG were crystallized in large-area single-crystal monolayers, which showed strongly polarized photoluminescence and demonstrated their high potential as active layers in solution-processed single-layer organic light-emitting transistors.